CARLSBAD, Calif., Sept. 24, 2019 /PRNewswire/ — Natural Alternatives International, Inc. (“NAI”) (Nasdaq: NAII), a leading formulator, manufacturer and marketer of customized nutritional supplements, today reported financial operating results for the fourth quarter and fiscal year ended June 30, 2019.
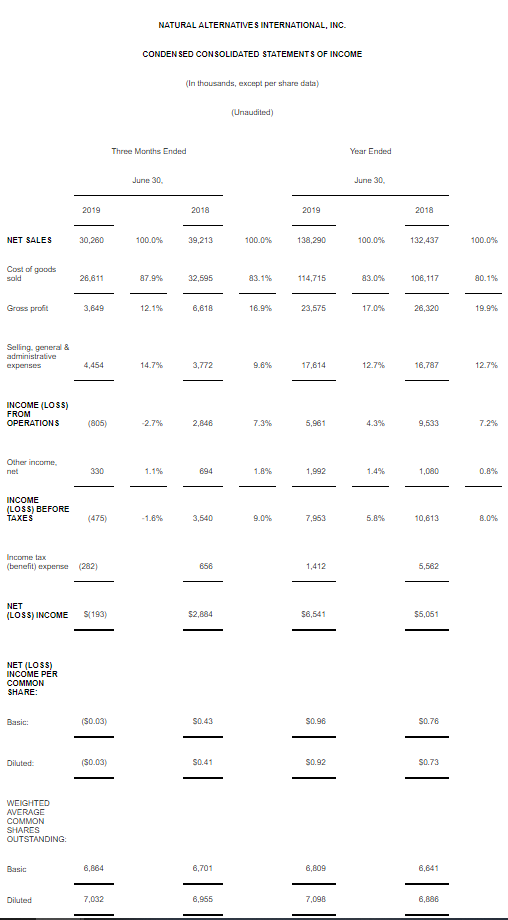
For the fiscal year 2019, net income was $6.5 million, or $0.92 per diluted share, on net sales of $138.3 million, compared to net income of $5.1 million, or $0.73 per diluted share, on net sales of $132.4 million for fiscal year 2018.
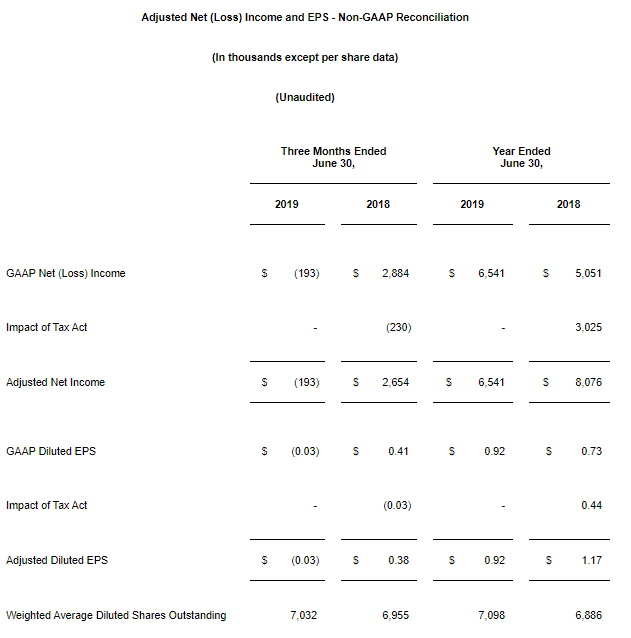
The previous period’s results were unfavorably impacted by one-time discrete tax expense amounts recorded in connection with the 2017 U.S. Tax Cuts and Jobs Act. These one-time charges totaled $3.0 million, or approximately $0.44 per diluted share. Excluding these discrete tax items, our adjusted net income during the fiscal 2018 was $8.1 million, or $1.17 per diluted share.
Net sales during fiscal year 2019 increased $5.9 million, or 4%, from $132.4 million recorded in the prior year. For fiscal year 2019, private-label contract manufacturing sales increased $10.6 million, or 10%, from the comparable period last year. The increase was due primarily to the sale of new products to new and existing customers, and higher volumes of current products to existing customers located primarily in the U.S. and Asian markets. Sales to our largest private-label contract manufacturing customer decreased 8.3%, however, sales to all other private-label contract manufacturing customers increased more than 40%. The sales decline from our largest private-label contract manufacturing customer primarily related to reduced demand in their international operations. Their sales decline accelerated during the fourth quarter of fiscal year 2019 and resulted in lower product re-orders.
CarnoSyn® beta-alanine royalty, licensing and raw material sales revenue decreased 22% to $16.7 million during fiscal year 2019 as compared to $21.4 million for fiscal year 2018. We believe this sales decline was impacted by certain customers discontinuing the use of our CarnoSyn® beta-alanine in favor of generic beta-alanine. We expect the recent ruling from the U.S. Court of Appeals for the Federal Circuit in favor of NAI’s CarnoSyn® beta-alanine patents, along with our CarnoSyn® beta-alanine New Dietary Ingredient (NDI) status from the FDA, will help motivate departed brands and contract manufacturers to purchase CarnoSyn® in the future. Additionally, we will continue to pursue all legal avenues to protect our CarnoSyn® brand, intellectual property portfolio, and regulatory compliance status.
Net sales during the three months ended June 30, 2019, decreased $9.0 million, or 23%, from $39.2 million recorded in the comparable prior year period. For the quarter ended June 30, 2019, private label contract manufacturing sales decreased $6.7 million, or 20%, from the comparable quarter last year. Sales to our largest private-label contract manufacturing customer declined over 50%, however, sales to all other private-label contract manufacturing customer increased over 50%, including sales of new products to new and existing customers.
CarnoSyn® beta-alanine royalty, licensing and raw material sales revenue decreased 42% to $3.2 million during the fourth quarter of fiscal year 2019, as compared to $5.4 million for the fourth quarter of fiscal 2018. We believe this sales decline was impacted by certain customers discontinuing the use of our CarnoSyn® beta-alanine in favor of generic beta-alanine.
We had a net loss of $0.2 million, or $0.03 per diluted share, for our fourth quarter of fiscal year 2019, compared to a net income of $2.9 million, or $0.41 per diluted share, in the fourth quarter of fiscal year 2018. This decrease was primarily attributable to a decrease in CarnoSyn® beta-alanine revenue, a one-time write off of $686,000 related to obsolete SR CarnoSyn® inventory, and increased legal and advertising expenses associated with our CarnoSyn® business, and decreased private-label contract manufacturing sales.
Given our planned business strategies, current currency headwinds and economic business trends, we expect private-label manufacturing sales growth in the low to mid- single digit percentage range in fiscal year 2020. This estimate includes expectations of flat to slightly down sales revenue for our largest private-label contract manufacturing customer.
Additionally, we expect CarnoSyn® and SR CarnoSyn® beta-alanine revenue growth in the mid-to high single digit percentage range during fiscal year 2020. However, based on our strategy to defend our patents, trademarks, and NDI status for our CarnoSyn® brands, the expansion of the Japanese marketplace for our CarnoSyn® brands, and increased marketing, advertising and promotion costs expected to be deployed for our launch into the Wellness and Healthy Aging markets for SR CarnoSyn®, we anticipate operating income will be flat to slightly down in fiscal year 2020 as compared to fiscal year 2019.
As of June 30, 2019, we had cash of $25.0 million and working capital of $57.1 million compared to $23.6 million and $50.9 million, respectively, as of June 30, 2018. As of June 30, 2019, we had $10.0 million available under our line of credit agreement.
Mark A. LeDoux, Chairman and Chief Executive Officer stated, “This final quarter of our fiscal year was challenging as we navigated some customer and regional softening of demand, primarily in Europe, and addressed inventory valuation challenges. Our team was able to limit these negative impacts, while focusing on new opportunities and expanding our penetration of other regional growth markets. The industry is experiencing the deleterious impacts of international supply chain tariffs, regional regulatory demands, which are often arbitrary or capricious, and uncertainties about potentially arduous new standards for products.”
“While these market conditions exist in the short term, having a strong balance sheet with no debt and sound levels of liquidity provides us a haven in an unpredictable environment. We continue to work with regulators globally to enforce statutory requirements, assuring product integrity with an ever vigilant eye to quality and safety of ingredients.”
“NAI was recently recognized by the venerable Natural Products Association for Innovation in Science, with its continued work in research surrounding CarnoSyn® and SR CarnoSyn® beta-alanine. This award is a significant acknowledgment of NAI’s industry leadership, commitment to innovation, research and development, and our perseverance to go above and beyond, including achieving NDI status from the FDA for CarnoSyn® beta-alanine.”
“While our recent results have fallen short of our expectations, we remain committed to expanding our profitability in this fiscal year through attention to detail across all business units. Additionally, we will continue to align our supply chain requirements in the most cost and quality effective manner available to our world-class production teams in the USA and Switzerland. Finally, we are committed to industry leadership with a steadfast commitment to quality.”
Reconciliation of Non-GAAP Information
The GAAP results contained in this press release and the financial statement schedules attached to this press release have been prepared in accordance with accounting principles generally accepted in the United States (“GAAP”). In addition to the Company’s results prepared in accordance with GAAP, the Company provided information on a non-GAAP basis. The manner in which this non-GAAP information is derived is discussed below, and the Company has provided in the tables to this release a reconciliation of the non-GAAP information to the most directly comparable GAAP information.
Net (Loss) Income and EPS Adjusted for Non-Recurring Tax Expense. In order to make the fiscal year 2018 results comparable to fiscal year 2019, we have presented net income and EPS for the fiscal 2018 on a non-GAAP basis by excluding discrete tax items related to the 2017 U.S. Tax Cuts and Jobs Act for the quarter and year ended June 30, 2018. The following is a schedule reconciling our Adjusted Net Income and EPS to our GAAP Net Income and EPS.
NAI, headquartered in Carlsbad, California, is a leading formulator, manufacturer and marketer of nutritional supplements and provides strategic partnering services to its customers. Our comprehensive partnership approach offers a wide range of innovative nutritional products and services to our clients including: scientific research, clinical studies, proprietary ingredients, customer-specific nutritional product formulation, product testing and evaluation, marketing management and support, packaging and delivery system design, regulatory review and international product registration assistance. For more information about NAI, please see our website at http://nai-online.com.
This press release contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934 that are not historical facts and information. These statements represent our intentions, expectations and beliefs concerning future events, including, among other things, future acquisitions, revenue profits and financial condition, our ability to maintain our patents, generate revenues from the commercialization of our patents and trademarks, secure compliance with our intellectual property rights, and develop, maintain or increase sales to new and existing customers, as well as future economic conditions and the impact of such conditions on our business. We wish to caution readers that these statements involve risks and uncertainties that could cause actual results and outcomes for future periods to differ materially from any forward-looking statement or views expressed herein. NAI’s financial performance and the forward-looking statements contained herein are further qualified by other risks, including those set forth from time to time in the documents filed by us with the Securities and Exchange Commission, including our most recent Annual Report on Form 10-K.
CONTACT – Michael Fortin, Chief Financial Officer, Natural Alternatives International, Inc., at 760-736-7700 or [email protected].
Web site: http://nai-online.com