Record Level of Sales, Net Income, and Diluted EPS for the Fiscal Year 2021
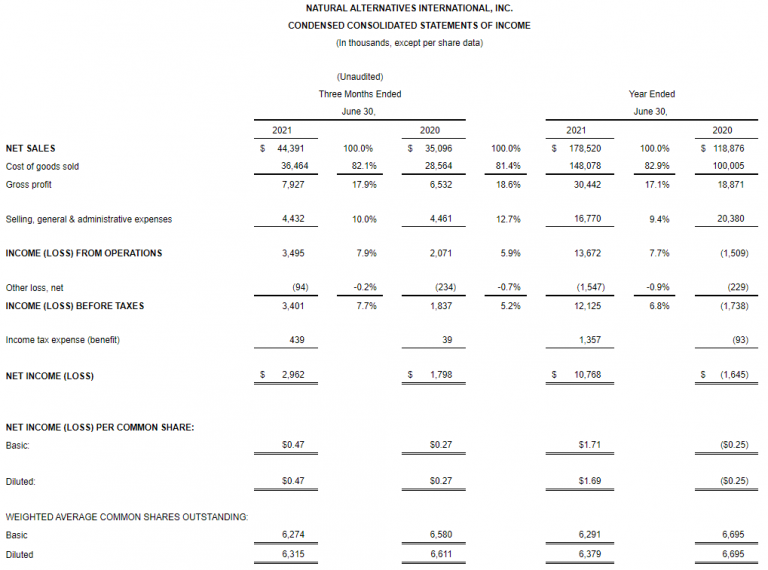
– 2021 Q4: $44.4 million Net Sales (+26.5%), $3.0 million Net Income, $0.47 diluted EPS
– Fiscal Year 2021: $178.5 million Net Sales (+50.2%), $10.8 million Net Income, $1.69 diluted EPS
– Appointment of Dr. Guru Ramanathan as a new independent Director
CARLSBAD, Calif., Sept. 20, 2021 /PRNewswire/ — Natural Alternatives International, Inc. (“NAI”) (Nasdaq: NAII), a leading formulator, manufacturer and marketer of customized nutritional supplements, today announced net income of $3.0 million, or $0.47 per diluted share, on net sales of $44.4 million for the fourth quarter of fiscal year 2021 compared to net income of $1.8 million, or $0.27 per diluted share, in the fourth quarter of the prior fiscal year.
Net sales during the three months ended June 30, 2021 increased $9.3 million, or 26.5%, to $44.4 million as compared to $35.1 million recorded in the comparable prior year period. During the same period, private-label contract manufacturing sales increased to $39.8 million, a 21.2% increase from the comparable quarter last year. In both the fiscal year and fourth quarter, private-label contract manufacturing sales increased primarily due to higher sales from a majority of our distribution channels worldwide. A significant portion of our increased contract manufacturing sales related to higher sales of immune and wellness products which is in line with the trend we find being experienced by the dietary supplement industry that appears to be driven by consumers taking a more active role in their health and wellness as a result of the COVID-19 pandemic. Our contract manufacturing sales also increased due to sales of newly awarded products from new and existing customers. CarnoSyn® beta-alanine royalty, licensing and raw material sales revenue increased 102.6% to $4.7 million during the fourth quarter of fiscal year 2021, as compared to $2.3 million for the fourth quarter of fiscal year 2020. In both the fiscal year and fourth quarter, CarnoSyn® sales increased primarily due to an increase in material shipments resulting from higher sales to existing customers. We believe the higher sales were influenced by an increase in activity as gyms and athletic facilities began to reopen in accordance with easing COVID-19 guidelines in various cities and states across the U.S.
Net income for the year ended June 30, 2021 was $10.8 million, or $1.69 per diluted share, compared to a net loss of $1.6 million, or $0.25 per diluted share, for the year ended June 30, 2020.
Net sales during the year ended June 30, 2021 increased 50.2% to $178.5 million as compared to $118.9 million recorded in the comparable prior year period. For the year ended June 30, 2021, private-label contract manufacturing sales increased 54.6% to $164.3 million as compared to $106.3 million during the comparable period last year. CarnoSyn® beta-alanine royalty, licensing and raw material sales revenue increased 12.9% to $14.2 million during the year ended June 30, 2021, as compared to $12.6 million for the year ended June 30, 2020.
Based on our current sales order volumes, and forecasts we have received from our customers, we anticipate our fiscal year 2022 consolidated net sales will increase between 5.0% and 10.0% as compared to fiscal year 2021. We also anticipate we will generate operating income between 7.0% and 9.0% of net sales for our fiscal year ending June 30, 2022. Sales and profitability during the first half of fiscal year 2022 are anticipated to decline when compared to the same period of fiscal 2021. Our expectations for the first half of fiscal year 2022 are being driven by continuing supply chain, labor and logistical constraints, all of which are expected to result in a backlog of existing orders that may not be delivered until the second half of fiscal year 2022. We currently anticipate these manufacturing challenges will be substantially resolved during the second half of fiscal year 2022. As a result, we expect sales and profitability in the second half of fiscal 2022 to exceed the comparable period in fiscal year 2021, with the overall fiscal year 2022 results reflecting an increase in both sales and profitability on a full year basis.
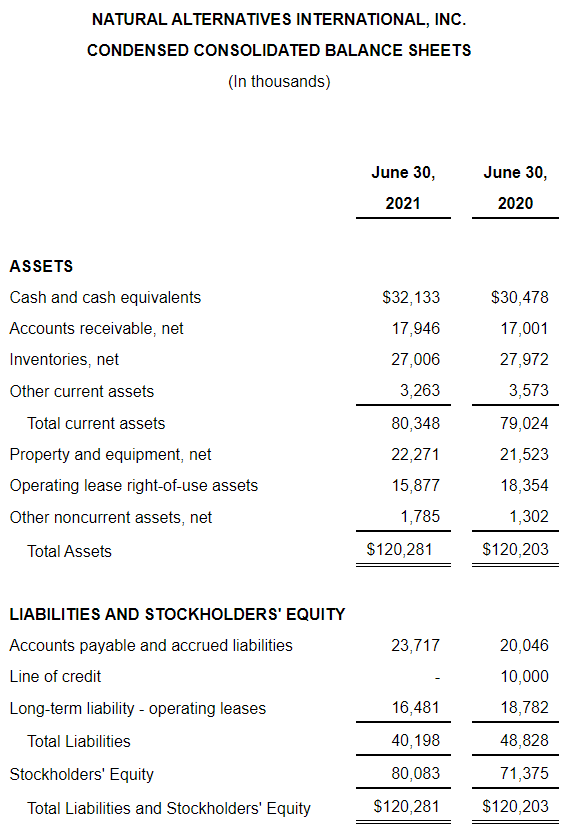
As of June 30, 2021, we had cash of $32.1 million and working capital of $58.3 million compared to $30.5 million and $51.2 million respectively, as of June 30, 2020. As of June 30, 2021, we had $20.0 million available under our line of credit agreement.
Additionally, on September 17, 2021, the Board of Directors appointed Dr. Guru Ramanathan to the Board of Directors seat vacated by the unexpected death of a director this past spring. Dr. Ramanathan joins NAI’s board with extensive experience in the dietary supplement industry including recently as Chief Innovation Officer with General Nutrition Corporation (GNC), and a founding member and current Chairman of the Supplement Safety & Compliance Initiative (SSCI). Dr. Ramanathan holds a Ph.D. from Tufts University in Healthcare Innovation Management, and an MBA from Duke University’s Fuqua School of Business.
Mark A. Le Doux, Chairman and Chief Executive Officer of NAI stated, “We are extremely proud of the fiscal year just concluded including record sales and profitability. With the recent acquisition of a new manufacturing and warehouse facility and the pending addition of a new blender in our current facility, we believe we are poised to take advantage of new opportunities that are at hand as well as future growth. Our balance sheet remains the envy of our industry and we are taking steps to continue leading by example in deploying production efficiencies, as well as securing essential raw materials and packaging components as they become available in a constrained supply environment.”
“Our recent acquisition of the new manufacturing facility in Carlsbad, California is being retrofitted to generate significant output of the highest quality whole-food-based meal replacement powders in various packaging configurations and other products envisioned for production in what will be a state-of-the-art cGMP facility. While it will likely be mid calendar 2022 or later before the facility is in full production, we anticipate this facility will allow us to significantly expand our product offerings across various industries and sales channels.”
“Our industry, like many, are dealing with the ongoing challenges of the COVID-19 pandemic, including supply chain and staffing, but we are fortunate that our industry has also benefited from consumers becoming more educated and interested in taking care of their health and well-being. We believe this bodes well for our growth objectives for the future. While we anticipate some challenges related to supply chain and staffing in the first half of this fiscal year, we believe we are well positioned to navigate these waters and we have made the appropriate investments in our business to ensure we are ready once those challenges clear.”
“We are extremely pleased to welcome Dr. Ramanathan to our board of directors. I have worked closely with Dr. Ramanathan for many years on various industry initiatives, including efforts by the Natural Products Association in Washington D.C. to facilitate the creation and launch of the retailer driven Supplement Safety Compliance Initiative (SSCI). Guru has a significant level of experience in dealing with mass market product positioning on a global basis. I believe he will be a valuable addition to our board and will provide a firsthand perspective with his extensive experience working on the branded side of our industry.”
An updated investor presentation will be posted to the investor relations page on our website later today (https://www.nai-online.com/our-company/investors/).
NAI, headquartered in Carlsbad, California, is a leading formulator, manufacturer and marketer of nutritional supplements and provides strategic partnering services to its customers. Our comprehensive partnership approach offers a wide range of innovative nutritional products and services to our clients including scientific research, clinical studies, proprietary ingredients, customer-specific nutritional product formulation, product testing and evaluation, marketing management and support, packaging and delivery system design, regulatory review and international product registration assistance. For more information about NAI, please see our website at https://www.nai-online.com.
This press release contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934 that are not historical facts and information. These statements represent our intentions, expectations and beliefs concerning future events, including, among other things, COVID-19 and related impacts on the availability of raw materials, and staffing, our future revenue profits and financial condition, our ability to develop, maintain or increase sales to new and existing customers, as well as future economic conditions and the impact of such conditions on our business. We wish to caution readers these statements involve risks and uncertainties that could cause actual results and outcomes for future periods to differ materially from any forward-looking statement or views expressed herein. NAI’s financial performance and the forward-looking statements contained herein are further qualified by other risks, including those set forth from time to time in the documents filed by us with the Securities and Exchange Commission, including our most recent Annual Report on Form 10-K.
Web site: https://www.nai-online.com
CONTACT – Michael Fortin, Chief Financial Officer, Natural Alternatives International, Inc., at 760-736-7700 or [email protected].